Table of Contents
I. What is HPMC?
The main features of HPMC:
Water-soluble non-ionic polymer;
Thermogel ability, solidification after heating;
It is biodegradable and biocompatible, suitable for daily chemical and pharmaceutical applications;
Excellent film-forming properties, widely used in coatings and adhesives.
II. HPMC structure: Chemical and molecular composition
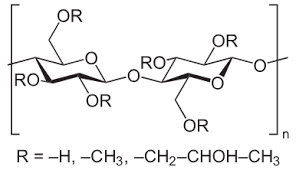
1. Overview of chemical structure
HPMC consists of a cellulose backbone and hydroxyl groups (-OH) partially substituted by methoxy groups (-OCH3) and hydroxypropyl groups (-CH2CHOHCH3).
Hydroxypropyl groups are highly hydrophilic and help HPMC disperse in water.
Methyl groups are highly hydrophobic and affect film-forming properties and thermal stability.
The performance of HPMC varies greatly depending on the ratio of these two groups.
These substituents determine its main properties:
Degree of Substitution (DS): The number of hydroxyl groups substituted by methyl groups.
Molar Substitution (MS): The average number of hydroxypropyl groups per glucose unit.
2. How structure affects properties
The higher the DS (degree of substitution), the stronger the hydrophobicity and the stronger the film-forming ability. Cold water solubility is reduced, but temperature resistance is better.
Higher MS (molar substitution) can increase water retention and solubility.
Balance DS and MS to optimize viscosity and gelation temperature.
3. Molecular weight and its impact
The molecular weight of HPMC ranges from tens of thousands to hundreds of thousands, which directly affects viscosity:
HPMC with higher molecular weight has higher viscosity and is suitable for thickening applications such as cement mortar or coatings.
Variants with lower molecular weight can be used in scenarios where rapid dissolution is required, such as pharmaceutical suspensions and ophthalmic solutions.
III. How HPMC structure affects properties
1. Water retention
2. Thickening and viscosity control
HPMC can be used as a rheology modifier to control the fluidity and consistency of paints, coatings and cleaners. Viscosity is determined by molecular weight and degree of substitution.
3. Film-forming properties
The balance of hydroxypropyl and methyl groups allows HPMC to form flexible films, such as tablet coatings that are not easy to break, and is also important in coatings.
4. Adhesion and processability
HPMC is able to improve the cohesion and open time of cement-based mortar, making it an important additive for building mortar and tile adhesives.

IV. HPMC structure in different applications
1. Construction industry
Wall putty and mortar: improve water retention and prevent cracking.
Self-leveling mortar: ensure smooth construction and extend open time.
Exterior insulation system (EIFS): helps improve adhesion and stability of insulation layer.
2. Pharmaceutical industry
Tablet coating: provides a smooth protective coating for controlled release.
Capsules: used for overall coating of capsules, which is not easy to become brittle under low moisture conditions.
Suspensions and ophthalmic solutions: ensure appropriate viscosity and stability.
Controlled release drugs: HPMC can be used in hydrophilic matrix tablets to adjust drug release time.
3. Paints and coatings
Used as a thickener to improve coverage and prevent pigment precipitation.
It has anti-sagging properties to ensure uniform application.
Enhance the adhesion and durability of coatings.
Improve rheological properties, improve flow and leveling.

4. Daily chemicals and detergents
Used as a thickener for shampoos, hand soaps and lotions.
Stabilizes formulations to prevent ingredient separation.
Enhances foam stability in detergents and shower gels.
Used as a binder and texture enhancer in toothpaste.
V. How to choose the right HPMC based on structure
Choosing the right HPMC grade depends on the following factors:
Viscosity: higher viscosity for construction applications and lower viscosity for pharmaceutical applications.
Substitution grade: optimized for specific properties such as water retention or film formation.
Degree of substitution: very important for food and pharmaceutical applications that require controlled thermal properties.
pH stability: different grades perform better in acidic or neutral conditions.
Compatibility with other ingredients: ensure optimal formulation in multi-component systems.




-scaled.jpg)








